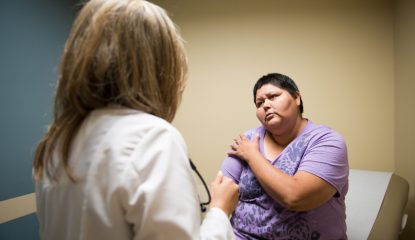Clinic Uses Predatory Tactics To Sell Costly Treatments To Desperate Patients
By Consumers For Quality Care, on December 6, 2019

A nationwide health clinic that has sold patients stem cell therapies for incurable lung conditions was scrutinized in a recent report in the Washington Post.
The company, Lung Health Institute (LHI) operates hundreds of clinics across the United States. The treatments they offer are very expensive and usually not covered by insurance. And, according to experts, there is little evidence they work.
The company’s descriptions of its procedure have evolved over the years, but the basic premise has remained the same: extract blood from patients, spin it in a centrifuge to isolate “stem cells” or “platelet-rich plasma platelet-concentrate,” then reintroduce those using an IV into the patient’s bloodstream, where they “naturally” find their way to damaged parts of the lung and heal them.
Lung disease experts say there is no evidence such treatments work.
The FDA has never approved the type of treatments that LHI offers, and it even considers some of them to be illegal.
The FDA has not approved most stem cell treatments and has said it considers many of them illegal. Miller and other officials at Lung Health Institute said they believe their treatment doesn’t require FDA approval. Nevertheless, the company now plans to apply to the FDA for approval, said Marc Scheineson, the company’s lawyer, “even though this is a long, expensive and arduous process … This action distinguishes LHI from the bulk of the other providers of stem cell and related blood-based therapies.”
The company has also been accused of aggressive selling tactics. If patients could not afford LHI’s expensive procedures, the company frequently pressured them to take on debt in order to pay. Some patients were encouraged to borrow money against their homes.
Former patients of the Tampa-based Lung Health Institute said they were encouraged to take out bank loans or borrow money from family members. Some withdrew from their retirement accounts and took up church offerings. Others borrowed against their homes.
LHI has aggressively marketed their products online, using search engines like Google and Bing.
Early on, the company’s marketers bought ads on search engines such as Google and Bing so its website would appear prominently whenever anyone searched for “cure” and “treatment” for illnesses such as chronic obstructive pulmonary disease, said three former marketing team members.
Although it continues to operate, the company has faced hurdles and scrutiny from the FDA and Google. In recent years, the FDA has made many of LHI’s former treatments illegal, and Google has recently announced new policies that bans ads for “unproven or experimental medical techniques such as most stem cell therapy.”